7 September 2012
Written by Gareth Powell

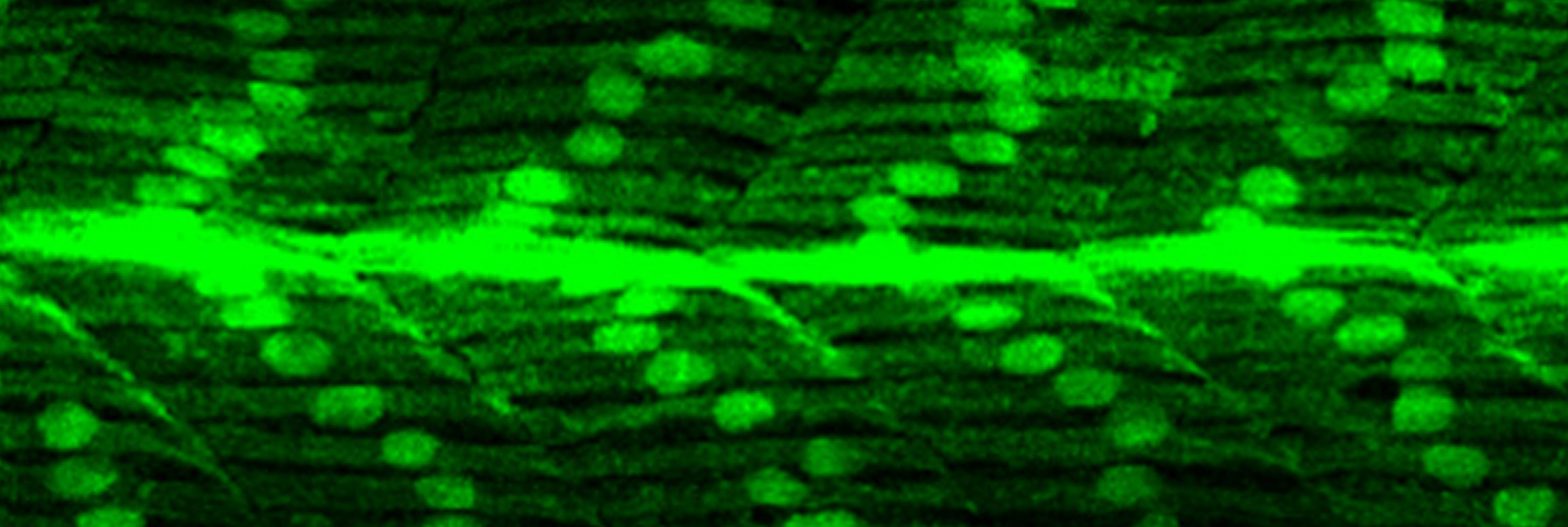
Embryonic neural and muscle precursor cells in a 16 hour old zebrafish embryo (cell periphery labelled in green). An individual muscle cell (highlighted in red on the right) must make a decision between founding a new muscle fibre or contributing to an existing one through cell fusion; just how this decision is made remains unknown.
Credit: Genome Research Limited
Muscles account for approximately 40% of our body weight (or more if you’re an Olympic athlete) and are essential for movement; they convert chemical energy into mechanical force to run a gold-medal winning race (or move your fingers to type this blog). There is a wide range of muscle diseases that hamper this ability. Malformation of the molecular machinery within muscles leads to diseases such as Duchenne muscular dystrophy, which is a result of weaknesses in the anchoring of muscle fibres, causing them to break and retract. Other muscle diseases are less well understood, such as sarcopenia, an age-related disease characterised by the loss of muscle mass, strength and function, and cachexia or “wasting syndrome” commonly seen in the advanced stages of cancer. Being able to boost the growth and repair mechanisms of muscle may be a very good way to treat at least some of these diseases.
However, we have a very important hurdle to overcome before we can start to treat muscle disease at the molecular level. We need a very detailed understanding of the biology of muscle. How it first forms in an embryo can give us insight into these processes.
Each muscle is composed of a bundle of long fibres packed with the protein filaments that contract together to shorten a muscle and generate force. Muscle is a fascinating tissue because each muscle fibre is formed by many precursor cells fusing together into a single elongated cell with many nuclei. There are very few examples of cell fusion in mammals: sperm and eggs, vital cells in the placenta and bone-remodelling cells all undergo fusion. Learning more about this process in muscle might also help us understand more about these tissues.
In the embryo, each individual muscle precursor cell has an important decision to make – to fuse to an existing fibre and make it bigger, or to start a new muscle fibre. How is this dilemma resolved? How is the balance between the number of fibres and the size and content of each fibre struck? There are two main possibilities – predetermination or random chance.
One possibility is a pre-patterning of the precursors into two groups before it all starts, as has been observed in fruitfly larvae. Another is random chance – a number of cells get together to form a muscle which varies in size or number around an average.
My recent work on zebrafish (Powell and Wright 2011, PLoS Biol. 9:e1001216) suggests that precursors are not subdivided, but follow a more sporadic model. The evidence for this is that when fusion is completely blocked in the embryo, each muscle tissue cell forms a new fibre – doubling the number of muscle fibres. Furthermore, the three essential cell fusion proteins known in zebrafish are expressed by all precursors, suggesting that they are not subdivided by specification.
In my article (Powell and Wright 2012, Trends Cell Biol. 22:391) I reviewed all the current evidence for or against the existence of a pre-patterned model in a vertebrate embryo. My opinion is that the weight of evidence is against subdivision of the precursors into two strict subgroups, but the jury is still out. Finding more essential genes for muscle tissue cell fusion will help us in resolving this question and discover just how those individual cells act together to form muscle.
Gareth Powell works in the Cell Surface Signalling Laboratory at the Wellcome Trust Sanger Institute and is interested in more...
Review Articles
Related Links:
Wellcome Trust Sanger Institute Cell Surface Signalling Laboratory






