

Image credit: Clothilde Chenal / Wellcome Sanger Institute.
Balancing life on and off the slackline, Dr Clothilde Chenal brings the same precision to her pioneering work in mosquito pangenomics, tracing how these disease vectors adapt, evolve and spread.
When Postdoctoral Fellow, Dr Clothilde Chenal first saw the advert for her current role at the Wellcome Sanger Institute, it felt uncannily specific. “It was written for me. I need to be there,” she remembers thinking. She got in her bright yellow Honda – affectionately called Logo – and drove all the way from Montpellier, France and across the channel to Cambridge. Clothilde brought not only her own wheels and slacklining equipment – a balance sport involving walking across suspended flat nylon fabric – but also her expertise gained during her doctoral research on malaria‐transmitting mosquitoes.
She is now a postdoc in Dr Mara Lawniczak’s group at the Sanger Institute, leveraging pangenomes to understand how mosquitoes adapt, diversify and spread disease. This work is a natural continuation of her doctoral research. During her PhD, generating new data on complex mosquito genomes, was difficult, so Clothilde had to be resourceful. She built her project around existing large‑scale datasets from the Sanger Institute and the MalariaGEN consortium.
In our latest blog, Clothilde shares with us some of the foundational work she has done in the world of mosquito genomics and explains how this has strengthened her present-day ability to contribute to mosquito pangenomics.
What is it like as a postdoc here at the Sanger Institute?
I really enjoy being at the Sanger Institute, and the team I’ve joined is wonderful. There are over 10 different individual countries represented in our group, which makes for really fun team potlucks and office chats.
The Sanger Institute feels like an environment where ambitious projects are possible; I appreciate being surrounded by people who are also working in fields where they are pushing for something new and creating change.
The city of Cambridge itself is a good place to be. It feels international and lively, and there are lots of opportunities to connect with people across disciplines. I have even found the ideal bakery, which I visit as often as possible. Any day starts better with good bread!
Where were you living before coming to the UK?
I came to the UK from France. France and England are not so far apart, so when moving here, I came with a bit more than a suitcase. My legendary, slightly rusty, yellow Honda – which was my grandfather’s car – came as well. All my life, I have known this car, and it has been part of all my major milestones. Moving to the UK, there was no question it would come with me. I packed this tiny car to the hilt! Coming across on the ferry, I felt ready. But then the driving was a bit worrying at first, as all the roundabouts and roads are all on the other side!

Catching the ferry to come to Britain in the legendary, slightly rusty, yellow Honda. Image credit: Clothilde Chenal / Wellcome Sanger Institute.
From France to the UK, you have kept mosquitoes as your main research area – why?
Mosquitoes are simply fascinating – from both a biological and a public health perspective. They are the most important transmitter of malaria, and they can adapt extremely quickly to what is around them (much quicker than I adapted to driving on UK roads or vinegar on chips!). That makes them challenging to study but also wonderful if you want to see evolution in action.
Do not tell the human geneticists – but in my opinion, mosquitoes are so much more interesting than humans. The way they change and adapt so quickly really caught my attention. I’m particularly interested in Anopheles funestus and the members of the Anopheles gambiae complex – a group of at least nine distinct species of mosquito; some of the species in this complex are capable of transmitting the malaria parasite.
It is very interesting that the members of the An. gambiae complex look identical; however, they can live in very different ecological niches. Some mosquito species have larvae that are able to develop in salty water, some in hot springs, some adult mosquitoes prefer humans, and some prefer cattle. They are an insect that really takes a serious all-you-can-eat buffet approach to hosts and habitats. This diversity and their flexibility make them a remarkable system for studying local adaptation, speciation and the spread of insecticide resistance. Although the species in the An. gambiae complex look-alike, we can use genomics to tease them apart.
How do you generate a pangenome, and what can it be used for?
A pangenome is a way of capturing all the genetic diversity in a species, instead of relying on a single reference genome from one or several specimens. I like to explain it as a map rather than a line. A single reference genome gives you one route through the genome, but a pangenome shows you all the possible paths.
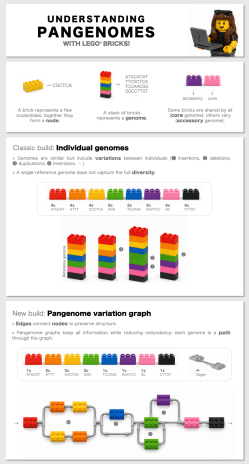
Why one reference genome isn’t enough: understanding pangenomes and genetic variation with LEGO® bricks. Genomes are often represented as a single reference sequence, but real populations and species contain much more diversity. Using a simple analogy, we show how pangenome graphs capture this variation while keeping all the information in a compact form. Image credit: Clothilde Chenal / Wellcome Sanger Institute.
I often use LEGO as a visual analogy. Each genome is like a pile of bricks, and although most of the structure is shared, each individual also has differences: insertions, deletions, inversions and duplications. A pangenome lets us represent all of those possibilities together. That matters because if we only rely on one linear reference genome, we can miss important variation that is not present in the individual we chose to sequence first.
I also think pangenomes are more than capturing individual diversity; a pangenome is a much better way of compressing and organising genomic data. For a place like the Sanger Institute, where the datasets are huge, the consideration of using pangenomes for data space and storage is a very practical advantage as well as a scientific one. Researchers using pangenomes still need standardisation and resources, infrastructure and leadership. I am part of a group of researchers around the world trying to change that!
The biggest promise of pangenomics is that it changes the way we think about genomes. A linear reference is useful, but it also creates bias. If a variant isn’t in the reference, it may become invisible during downstream analyses. That matters a lot in mosquitoes, where variation can affect public health and safety measures such as insecticide resistance, host preference, ecology and potentially even speciation. I want to use pangenomics to make that diversity visible and to connect genomic structure with evolutionary and epidemiological questions. I also hope that, over time, pangenomes become the default rather than the exception. If we can include diversity from the beginning, we will get a more accurate picture of biology.
What are the different uses for short-read and long-read sequencing in your research?
During my doctoral research, I wanted to understand how mosquitoes adapt locally and how they begin to diverge into different species, but I had to work with short-read data. Short reads are a way to build a sharper picture of a genome, but they can have issues due to repeat regions of DNA all looking the same in small chunks and being thrown out as ‘junk’. Short-read DNA sequencing struggles with repetitive regions of DNA, especially in transposable elements, which are bits of DNA that jump around. Often, transposable elements are really exciting instances for evolution. So, to avoid missing out on these, I naturally became interested in long-read sequencing.
Long-read sequencing provides an output of the genome in much bigger chunks of DNA, which is easier to assemble but doesn’t give as crisp an image of the genome as short-read sequencing. For pangenomes, we really want the best of both worlds – both short and long reads – to build a diverse picture of a species’ genome.
Now, at the Sanger Institute, I am working to go beyond the limitations of short-read DNA by generating long-read data for mosquitoes. That lets me build much richer pangenomes and capture parts of the genome that were previously very difficult to study.
Left: Mosquito larvae being grown in the Sanger Institute insectarium. Right: Adult mosquitoes living on sugar water. Videos credit: Carmen Denman Hume / Wellcome Sanger Institute.
What is your project at the Sanger Institute focussed on now?
I am working on An. gambiae complex and An. funestus mosquito genomes – both are able to transmit malaria. At the Sanger I am building pangenomes for these mosquitoes across several locations where they are found. Soon, I will receive more data from collaborators that will include samples from across Africa. I will use these genomes to answer biological questions about malaria-carrying mosquitoes – all the while troubleshooting technical challenges related to handling these hugely complex datasets.
You have developed the tools required to answer research questions – what is next?
To build a community. Pangenomics is such a fast-moving field. The concept has been around for a while, but the tools we use now have really only developed over the last decade or so. A lot of those tools were originally created for bacteria or for humans, and they don’t always transfer well to highly diverse mosquito genomes.
That means there is still a lot of work to do around standardisation. Every month, there seems to be a new tool or pipeline, and although that’s exciting, it can also be difficult to keep up with. To help keep up, it is so important that people from different communities talk to one another. Here at the Sanger Institute, I am involved in organising a monthly pangenomes meeting, where attendees discuss their own work or new papers in the field. This has been really great to build collaborations and find others working on the same challenges.

Clothilde at work in her office. Image credit: Carmen Denman Hume / Wellcome Sanger Institute.
Amidst the busy life of a postdoc, what do you do to relax?
There is one thing that focusses my mind like nothing else: slacklining. It is a sport that demands strength and focus. When I am walking on the slackline, whether it is a few inches off the grass here in Cambridge or walking between cliff edges, each movement you make creates or destroys your balance and momentum. Every vibration you feel, you have personally created. You must tune into how your body feels and moves. For me, this is totally a release from any work stress. Of course, it can look risky but can be done very safely. Anyone interested in giving it a try, I always have my slackline in the boot of my yellow Honda!
Clothilde slacklining in her spare time. Image credit: Clothilde Chenal / Wellcome Sanger Institute.








